Dẫn từ từ khí CO2 vào dung dịch hỗn hợp gồm NaOH và Ba(OH)2. Sự phụ thuộc của khối lượng kết tủa (y gam) vào số mol CO2 (x mol) được biểu diễn bằng đồ thị sau:
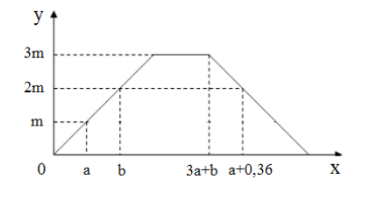
Giá trị của m là
A. 17,73. B. 7,88. C. 14,184. D. 11,82.
Hãy suy nghĩ và trả lời câu hỏi trước khi xem đáp án
Lời giải:
Báo saiĐoạn 1: CO2 + Ba(OH)2 → BaCO3 + H2O
\(% MathType!MTEF!2!1!+- % feaahqart1ev3aqatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeaacaGaaiaabeqaamaabaabaaGcbaGaeyOKH4Qaam % yyaiabg2da9maalaaabaGaamyBaaqaaiaaigdacaaI5aGaaG4naaaa % caGG7aGaamOyaiabg2da9iaaikdacaWGHbGaai4oaiaad6gadaWgaa % WcbaGaamOqaiaadggadaqadaqaaiaad+eacaWGibaacaGLOaGaayzk % aaWaaSbaaWqaaiaaikdaaeqaaaWcbeaakiabg2da9iaaiodacaWGHb % aaaa!4BB4! \to a = \frac{m}{{197}};b = 2a;{n_{Ba{{\left( {OH} \right)}_2}}} = 3a\)
Khi \(% MathType!MTEF!2!1!+- % feaahqart1ev3aqatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeaacaGaaiaabeqaamaabaabaaGcbaGaamOBamaaBa % aaleaacaWGdbGaam4tamaaBaaameaacaaIYaaabeaaaSqabaGccqGH % 9aqpcaaIZaGaamyyaiabgUcaRiaadkgacqGH9aqpcaaI1aGaamyyai % abgkziUkaad6gadaWgaaWcbaGaamOqaiaadggacaWGdbGaam4tamaa % BaaameaacaaIZaaabeaaaSqabaGccqGH9aqpcaaIYaGaamyyaiaacU % dacaWGUbWaaSbaaSqaaiaadkeacaWGHbWaaeWaaeaacaWGibGaam4q % aiaad+eadaWgaaadbaGaaG4maaqabaaaliaawIcacaGLPaaadaWgaa % adbaGaaGOmaaqabaaaleqaaOGaeyypa0JaaG4maiaadggacqGHsisl % caaIYaGaamyyaiabg2da9iaadggacaGG7aGaamOBamaaBaaaleaaca % WGobGaamyyaiaadIeacaWGdbGaam4tamaaBaaameaacaaIZaaabeaa % aSqabaGccqGH9aqpcaaIYaGaamyyaaaa!64FD! {n_{C{O_2}}} = 3a + b = 5a \to {n_{BaC{O_3}}} = 2a;{n_{Ba{{\left( {HC{O_3}} \right)}_2}}} = 3a - 2a = a;{n_{NaHC{O_3}}} = 2a\)
Bảo toàn \(% MathType!MTEF!2!1!+- % feaahqart1ev3aqatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeaacaGaaiaabeqaamaabaabaaGcbaGaam4qaiabgk % ziUkaadggacqGHRaWkcaaIWaGaaiilaiaaiodacaaI2aGaeyypa0Ja % aGOmaiaadggacqGHRaWkcaaIYaGaamyyaiabgUcaRiaaikdacaWGHb % aaaa!4504! C \to a + 0,36 = 2a + 2a + 2a\)
\(% MathType!MTEF!2!1!+- % feaahqart1ev3aqatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeaacaGaaiaabeqaamaabaabaaGcbaGaeyOKH4Qaam % yyaiabg2da9iaaicdacaGGSaGaaGimaiaaiEdacaaIYaGaeyOKH4Qa % amyBaiabg2da9iaaigdacaaI5aGaaG4naiaadggacqGH9aqpcaaIXa % GaaGinaiaacYcacaaIXaGaaGioaiaaisdaaaa!49DE! \to a = 0,072 \to m = 197a = 14,184\)
Đề thi thử tốt nghiệp THPT QG môn Hóa năm 2020
Trường THPT Hậu Lộc 4 Thanh Hóa lần 2